Definition of the events to be logged
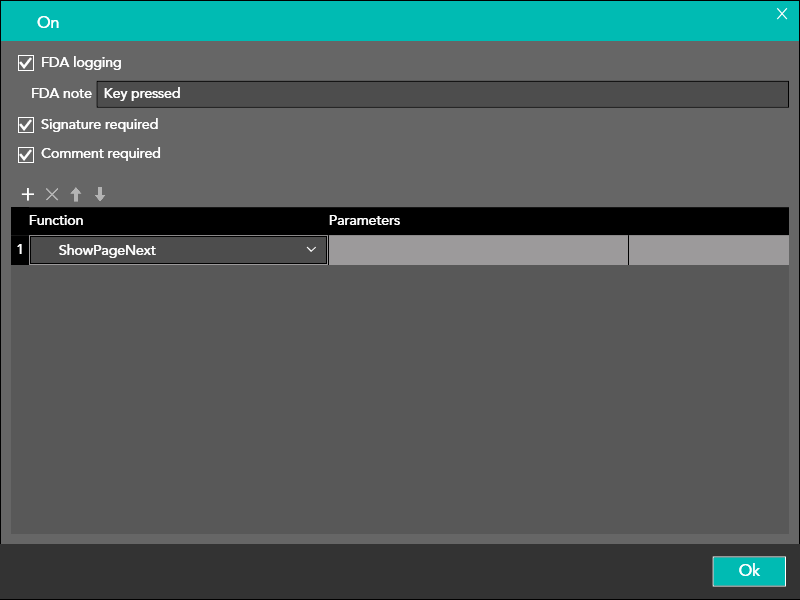
The user can select which events are to be tracked in the FDA log by selecting the FDA Logging option in the event schedule window.
It is also possible to enter the following parameters:
|
FDA Note
|
Custom text saved in the logfile
|
|
|
Signature required
|
The electronic signature has to be entered before moving ahead with the functionality
|
The electronic signature is not a login parameter but will be recorded in the event log buffer
|
|
Comment required
|
The user has to enter a comment before moving ahead with the functionality
|
|
The function that must be performed when the event occurs is inserted in the lower part of the window.
Tag editing mode
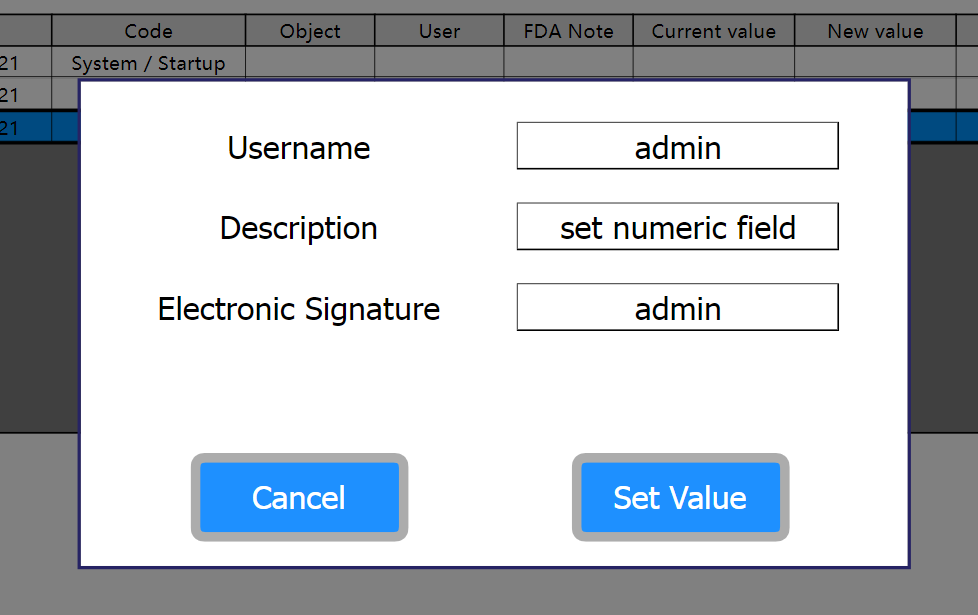
The specification provides that at the end of the insertion of the new value of a tag the following information is recorded in the log buffer:
-
User name
-
Description of the reason why the Tag value has been modified
-
Electronic signature
Buffer di Log
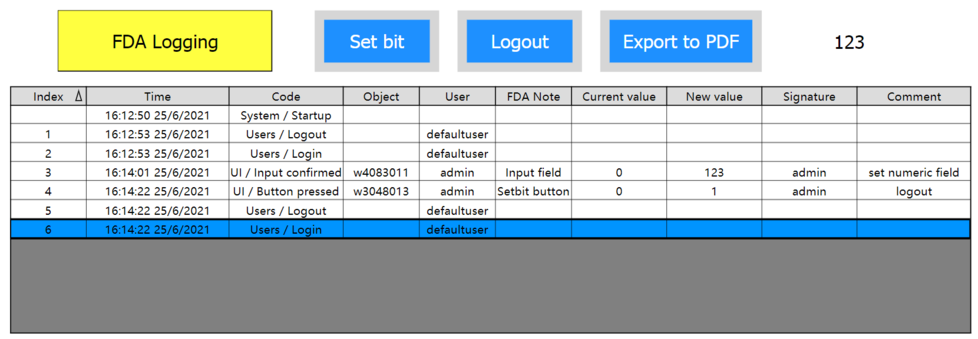
The log buffer will be displayed in the event system widget
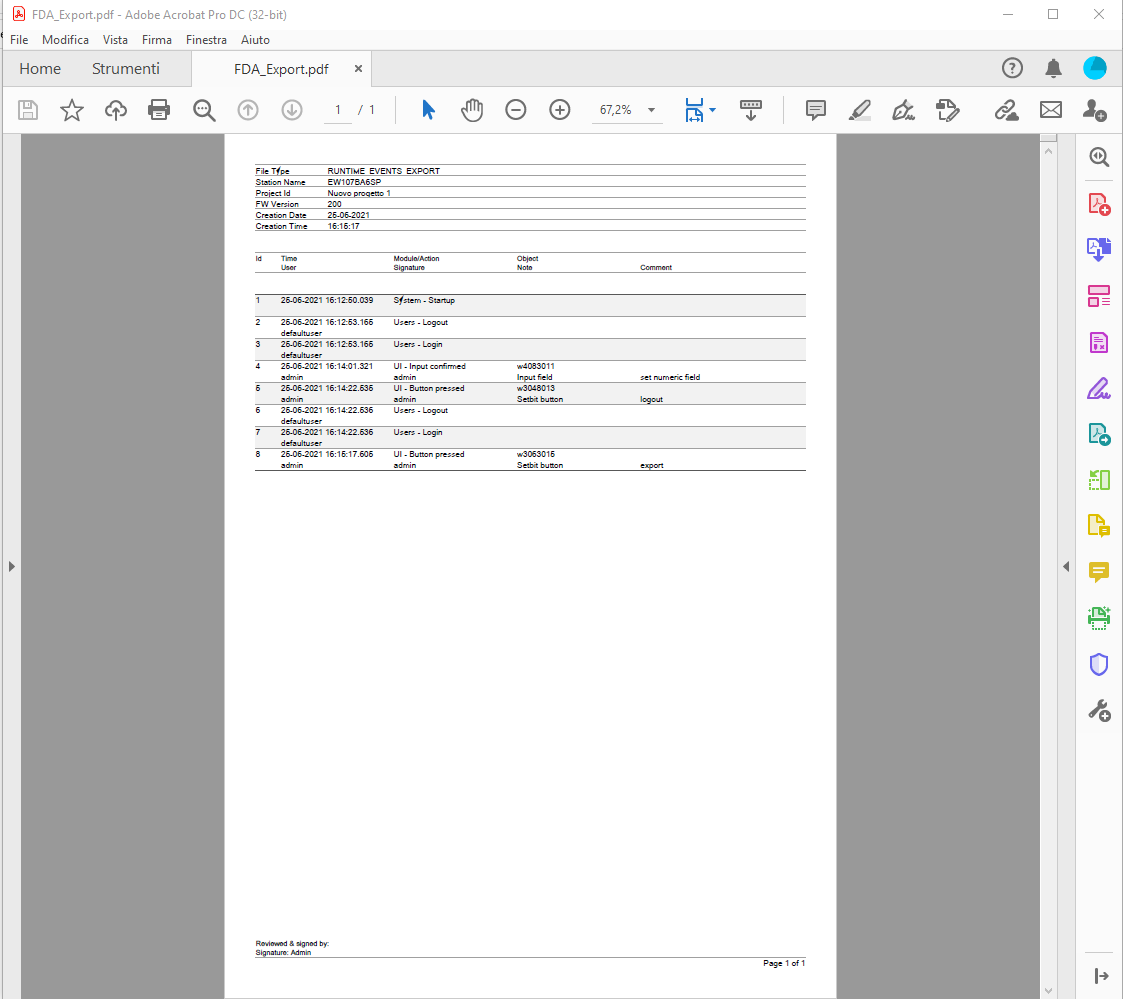
Logbuffer exporting
The log buffer can be exported as below:
-
CSV file
-
PDF (with or without electronic signature)
-
Database table